Summary: Compare home-use PEMF mats by operating data only – measurable device descriptors such as magnetic flux density, frequency, waveform disclosure, control model, coil transparency, and regulatory wording. These details describe how a device is specified, but they do not prove treatment value, safety outcomes, or superiority for any condition.
Shopping for a PEMF mat means wading through specification sheets filled with numbers, labels, and claims that look technical but often blend measurable operating data with promotional language. The difference between the two matters more than most buyers realize. A Gauss number, a frequency range, or an FDA-related phrase can describe a device accurately – or it can be stretched into unsupported inference about what the device actually does for your body.
This article teaches you how to read those specifications as device descriptors: what they measure, what they mean for comparison, and where they stop being evidence of anything beyond hardware design.
PEMF Advisor publishes buyer-oriented reviews, comparisons, and educational content for home-use PEMF mats and devices. This page is a constrained claim-literacy explainer within that system: it focuses only on how buyers should interpret PEMF specification claims more carefully, not on diagnosis, treatment guidance, protocol recommendations, or condition-based product selection.
If you want the broader specification context around frequency, intensity, waveform, controls, and disclosure quality, use our PEMF Specs & Controller Explained guide. This page supports that larger framework by focusing on one narrower question: how to read PEMF spec claims without turning device descriptions into unsupported treatment logic.
Spec / Claim Pattern Quick Table
PEMF specification literacy starts with operating data, not with benefit language. A spec can be measured, compared, or disclosed without proving outcomes. Before diving into the details, the table below separates measurable operating data from unsupported interpretation at a glance.
What This Table Is For
Think of this table as a decoding tool, not a verdict tool. Its purpose is to stop you from mistaking disclosure for proof. The table compresses the main contrast between measurable specs and unsupported interpretation into a single scannable reference. Observed claims in the PEMF market often correlate waveforms or presets with healing potential, but those inferences are not stable buying proof.
Use this as your first filter before you evaluate any product page.
The Fastest Reading Rule for Any PEMF Spec Page
Read every PEMF spec in three steps: first ask what the claim literally measures, then ask under what conditions it was measured or disclosed, and only then decide whether it improves comparison quality. If the page jumps from a number or label straight into a promised effect, that is the point where description has started turning into interpretation.
This rule matters because most buyer confusion does not begin with false numbers. It begins when a real number is treated as if it automatically explains what the device will do in the body. The purpose of this page is to stop that jump before it shapes the buying decision.
Spec / Claim Pattern – What It Measures – What It Means – What It Does NOT Prove
| Spec / Claim Pattern | What It Measures | What It Means for the Device | What It Does NOT Prove |
| Magnetic Flux Density (Gauss / Tesla / µT) | Field strength at a stated point | Describes how strong the magnetic field is under specific measurement conditions | Does not prove treatment efficacy, deeper penetration, or superior biological effect |
| Frequency (Hz) | Pulses per second | Describes the pulse rate of the device’s operating cycle | Does not prove condition-specific targeting or disease-relevant dosing |
| Waveform Geometry (Square, Sine, Sawtooth) | Pulse shape labeling | Describes the rise and fall pattern of each pulse | Does not prove that one shape produces superior biological responses |
| Presets vs. Manual Control | Interface and parameter access | Describes whether settings are locked into programs or adjustable by the user | Does not prove healing potential or clinical superiority |
| Coil Configuration | Hardware layout transparency | Describes the number, size, material, and arrangement of induction coils | Does not prove better outcomes or field-quality superiority |
| FDA Registered | Administrative listing status | Indicates the manufacturer listed the device with the FDA as a General Wellness product | Does not prove disease-specific efficacy or medical approval |
| FDA Cleared | Regulatory review outcome | Indicates the device passed a specific regulatory review pathway (such as 510(k)) | Should not be collapsed with “FDA Registered” – different regulatory status |
| “Medical Grade” | Nothing measurable in this category | A marketing descriptor, not a technical PEMF specification | Does not function like Gauss, Hz, waveform, or coil disclosure |
| Heat / Stones / Crystals / Layered Materials | Bundled non-PEMF product features | Describes materials or supplementary technologies included in the mat | Does not replace or improve core PEMF operating data |
Key Distinction
Disclosure quality is a comparison signal, not proof of results. A product page that openly states field strength, frequency behavior, and coil layout gives you more to compare – but that transparency alone does not validate health outcomes.
What Counts as Operating Data on a PEMF Product Page
Operating data is the only reliable comparison field when evaluating home-use PEMF mats. It includes any measurable or directly disclosed device descriptor that you can compare across products without importing inference about what the device does to the body. Operating data is constrained by measurement conditions and disclosure conditions, which means the context behind a number matters as much as the number itself.
Define Operating Data in Buyer Language
Operating data means the measurable or directly stated settings and hardware descriptors a buyer can compare across products. These include field strength (measured in Gauss, Tesla, or microTesla), pulse rate (measured in Hz), waveform labeling (square, sine, sawtooth), control model (presets vs. manual), and hardware transparency (coil configuration).
Operating data differs from claims about what a device supposedly does to the body. When a product page says “3000 Gauss,” that is operating data. When it says “penetrates deep tissue for cellular repair,” that is interpretation layered on top of data – and the two should not be confused.
Quick Definition
Operating data = what the device is specified to do electrically and mechanically. Not what the seller says it does biologically.
Why Operating Data Is the Only Reliable Comparison Field
Operating data enables direct comparison across products because it describes hardware behavior in shared terms. Regulatory or marketing language can shape perception without improving comparability. A phrase like “medical-grade pulsed therapy” sounds authoritative, but it adds nothing you can measure or compare side by side.
Transparency helps readers judge what is actually being disclosed. If one manufacturer publishes field strength at multiple distances, coil count, and frequency range while another only says “powerful PEMF technology,” the first gives you more comparison material. Disclosure quality is useful because it reveals how much real device information is available – but disclosure itself is not evidence of outcomes.
Hybrid features (heat layers, crystals, far-infrared) and promotional labels (“NASA-proven,” “biocompatible”) belong outside the core operating-data layer. They may describe real product features, but they do not function as PEMF specifications.
The Difference Between a Strong Spec and a Strong Claim
A strong spec is one that can be measured, stated clearly, and compared under visible conditions. A strong claim, by contrast, is often just a persuasive sentence built around that spec. The two can appear next to each other on the same product page while doing very different jobs.
For buyers, this means the technical-looking part of a sentence should not be allowed to carry the whole sentence’s meaning. “3000 Gauss at the mat surface” is a specification. “3000 Gauss for deep cellular repair” is a specification joined to an outcome claim. Only the first half belongs to the comparison layer.
What Happens When Buyers Overread PEMF Spec Claims
Overreading turns a device descriptor into unsupported treatment logic. Buyers often move from “this is disclosed” to “this works better,” even when that jump is not justified by the specification itself. A single number, label, or preset list can distort comparison when it is read as outcome evidence rather than as a hardware description.
Claim pattern literacy is the defense against this. When you encounter a number or label on a product page, the question is not “what does this do for me?” but “what does this tell me about the device?” The boundary between descriptive reading and prohibited condition-led interpretation is exactly where most shopping mistakes happen.
Common Overread Pattern
Descriptive reading: “This mat operates at 10 Hz.”
Overread: “10 Hz is the frequency that heals bone fractures.”
The spec describes a pulse rate. The overread imports an unverified treatment claim.
How to Read the Core PEMF Specs as Device Descriptors
The main PEMF specifications – magnetic flux density, frequency, waveform geometry, control model, coil configuration, and timer logic – are device descriptors. They tell you what the hardware is designed to do electrically. None of them proves outcomes on its own.
What a Spec Can Describe but Not Conclude
A PEMF specification can describe a device’s operating behavior without concluding what that behavior means biologically. That is the core reading discipline buyers need. A number may be real, disclosed, and comparison-useful while still being incomplete as evidence for anything beyond hardware design.
This is especially important in PEMF because the market often compresses multiple layers into one sentence: operating data, interpretive language, and outcome suggestion. Reading the spec correctly means separating those layers before you compare products.
Magnetic Flux Density: What a Gauss Number Measures
Intensity is the measure of magnetic field strength. On PEMF product pages, it is expressed in Gauss, Tesla, or microTesla. Magnetic flux density varies by distance from the coil and is constrained by coil design, which means a Gauss number describes field strength under stated (or sometimes unstated) measurement conditions.
A headline number without context is incomplete. “3000 Gauss” measured at the coil surface means something different from “3000 Gauss” measured three inches away. The later section on the inverse square law explains why this matters.
| Unit | Abbreviation | Relationship |
| Gauss | G | 1 Gauss = 100 microTesla (µT) |
| Tesla | T | 1 Tesla = 10,000 Gauss |
| microTesla | µT | 1 µT = 0.01 Gauss |
Frequency: What Hz Measures on a PEMF Device
Frequency refers to the number of pulses delivered per second and is measured in Hertz (Hz). On a PEMF device, frequency is constrained by the control model – some devices operate at a single fixed frequency, while others sweep across a stated range.
A fixed frequency and a sweeping range are different types of disclosure. A device listed at “10 Hz” runs at one pulse rate. A device listed at “1–50 Hz” moves across a range, either automatically or through user selection. Both describe operating behavior. Neither describes disease targeting.
Condition-linked frequency claims (e.g., “this frequency targets inflammation”) are low-stability marketing patterns. They should not be treated as stable buying proof under any reasonable reading of the specification.
Waveform Geometry: What the Pulse Shape Label Is Describing
Waveform geometry refers to the rise and fall shape of the pulse. The common labels – square wave, sine wave, and sawtooth wave – are device-description terms constrained by electronics design. They tell you what shape the electrical pulse takes, not what the pulse accomplishes biologically.
Some sources claim one waveform shape is more effective than others. Under these constraints, that claim remains low-stability or governance-restricted. The spec itself stops at pulse-shape description.
Proprietary waveform labels can obscure comparison when they are not translated into standard terms. If a manufacturer uses a branded name like “OmniWave” or “BioFlux Pulse,” you cannot compare it to a square or sine wave unless the underlying geometry is disclosed.
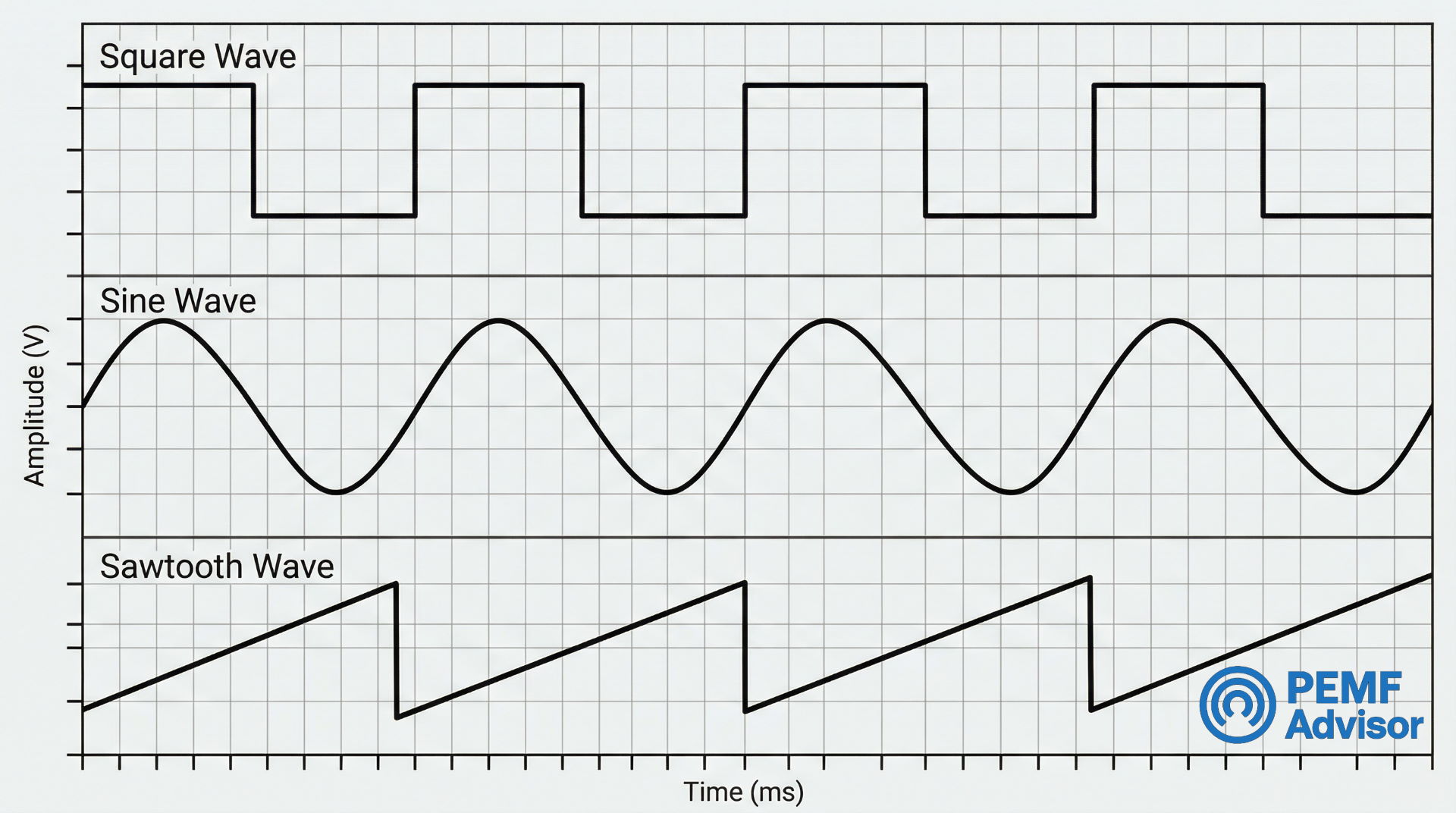
| Standard Label | What It Describes | Proprietary Label Risk |
| Square wave | Sharp rise, flat top, sharp drop | May be relabeled without changing the underlying shape |
| Sine wave | Smooth, continuous rise and fall | Branded names can hide a standard sine geometry |
| Sawtooth wave | Gradual rise, sharp drop (or inverse) | May be bundled into composite programs without clear labeling |
Presets vs. Manual Control: What the Controls Actually Tell You
Preset vs. manual control describes the capability to change parameters versus using hardcoded programs. A preset-only device runs pre-loaded routines. A device with manual control lets you adjust individual parameters like frequency or intensity directly.
More presets do not automatically mean better disclosure or better outcomes. In some cases, a large number of presets actually reduces transparency – the operating logic is hidden behind marketing labels like “Sleep Program” or “Recovery Mode” without revealing the underlying frequency, intensity, or waveform settings.
The more useful question is whether the device discloses the actual operating parameters behind its programs or locks them away from the buyer.
| Feature | Presets | Manual Control |
| Parameter access | Locked into pre-loaded programs | User can adjust frequency, intensity, or both |
| Transparency | Settings may be hidden behind program names | Actual operating values are visible |
| Comparison value | Hard to compare across brands if logic is opaque | Easier to compare when parameters are stated |
Coil Configuration: What Hardware Transparency Adds to Comparison
Coil configuration covers the number, size, material, and arrangement of induction coils inside the mat. It enables field-generation pattern disclosure, which helps readers judge how much operating detail is actually being shared.
When a manufacturer discloses coil count, coil material (e.g., copper vs. aluminum), and arrangement pattern, you can compare that detail against competitors. When coil disclosure is missing entirely, you lose a meaningful layer of comparison. Hardware transparency is a trust signal for the comparison process, not a proof signal for health outcomes.
Timer Logic and Basic Operating Controls
Timer logic enables session-length automation. Auto-shutoff intervals and basic controls describe usability, not efficacy. Whether a mat runs for 20 minutes or 60 minutes before shutting off is a convenience feature that matters for ownership experience without becoming a proof claim about results.
Timer and control features belong in the supporting-attribute tier – worth noting during comparison, but not a primary spec anchor.
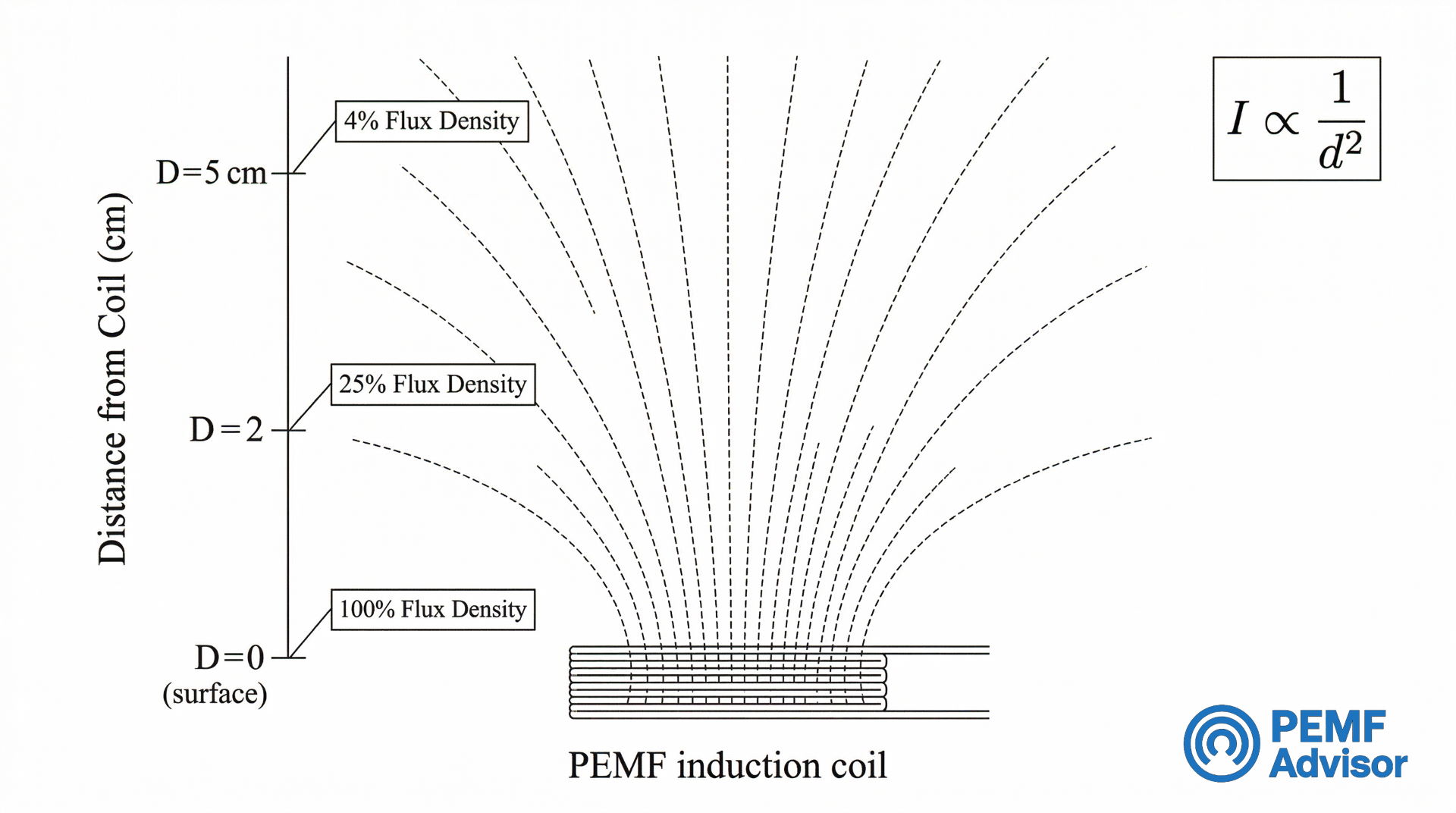
Why Distance Changes the Meaning of Intensity Claims
Magnetic flux density varies by distance from the coil. PEMF intensity diminishes rapidly as distance increases, and buyer interpretation is constrained by this physical reality. Surface Gauss claims vary by measurement location, and operating data is constrained by measurement conditions. Without understanding this, two seemingly identical Gauss numbers can describe very different field behaviors.
Surface Value vs. Value at Distance
A surface value is not the same as a value measured farther from the coil. Buyers should not assume the top-line Gauss number stays constant through space. Measurement location is part of the spec’s meaning. A mat advertised at 3000 Gauss measured at the coil surface may deliver a fraction of that strength even a few centimeters away. The headline number describes a peak at one point, not a uniform field across the mat’s usable area.
The Inverse Square Law as the Missing Reading Rule
Field strength drops quickly as distance from the source increases. This principle – roughly described by the inverse square law – is the single most important reading rule for evaluating Gauss claims, and it is almost never mentioned on product pages.
In plain terms: if a field measures a certain strength at the coil surface, it will be substantially weaker just a short distance away. The rate of drop-off depends on coil design, but the direction is always the same – away from the source means less intensity. This is a buyer-reading rule, not a treatment recommendation.
Why “Up to X Gauss” Is Incomplete Without Measurement Context
“Up to X Gauss” does not tell you where or how the value was measured. A single maximum figure can omit field distribution and distance context entirely. Two products advertising “up to 3000 Gauss” may have very different field behaviors if one was measured at the coil and the other at the mat surface after material layers.
Incomplete disclosure formatting can make two products look comparable when they are not. When reading intensity claims, ask: measured where? Peak or average? At contact or at distance?
Bigger Numbers Become Weaker Claims When Context Is Missing
A large intensity number can look more persuasive than a smaller one, but missing context usually weakens its comparison value rather than strengthening it. A product that publishes a lower value with clear distance, surface, and reporting-style disclosure often gives the buyer more usable information than a product that leads with a maximum figure and leaves the measurement frame unstated.
This is one of the main anti-hype reading rules for PEMF product pages: a bigger number without its conditions is often a weaker comparison claim than a smaller number with its conditions fully stated.
Red Flags for Intensity Claims
- Single “up to” number with no measurement location stated
- No distinction between peak and average values
- No mention of distance from the coil
- No disclosure of whether the value applies to the mat surface or the coil itself
Peak, Average, and Unspecified Intensity Numbers
A stated intensity can refer to a peak value (the highest momentary reading), an average value (the mean across a cycle), or an unspecified value where the manufacturer does not clarify which type it is. Missing qualification reduces clarity and makes product-to-product comparison less reliable.
| Intensity Type | What It Describes | Comparison Risk When Unspecified |
| Peak | The highest momentary field strength during a pulse cycle | May overstate the typical operating strength |
| Average | The mean field strength across the full cycle | May understate the peak output |
| Unspecified | No clarification on which measurement is reported | You cannot reliably compare it against a product that specifies peak or average |
Where PEMF Spec Claims Get Overinterpreted
Several common patterns in the PEMF market push buyers from descriptive reading into unsupported inference. Recognizing these patterns is the core of claim pattern literacy.
The Intensity War: High Gauss vs. Low-Intensity Resonance
The market often presents conflicting messages about intensity. High-intensity systems claim that stronger fields are needed. Low-intensity systems claim that resonance at lower power is the real key. Both positions are spec-positioning frameworks used in marketing – not proof frameworks supported by the specification itself.
The stable, compliant reading is to treat both as what they are: descriptions of where a device sits on the intensity spectrum. The distinction between high-intensity and low-intensity systems can be described technically (e.g., milliGauss vs. thousands of Gauss) without assigning outcomes. The contradiction between them is itself a reason to avoid overinterpreting intensity numbers.
Waveform Superiority Claims and Why They Outrun the Spec Itself
Waveform labels describe geometry, not superiority. Some sources claim one waveform shape improves biological effects more than another. Under these constraints, that claim remains low-stability. The spec itself stops at pulse-shape description – it tells you the shape of the signal, not what the shape does inside the body.
When a product page says “square waves penetrate deeper” or “sine waves are more natural,” it has moved from device description to biological assertion. That is the point where the specification can no longer support the claim.
Why More Presets Do Not Prove Greater Healing Potential
A larger number of presets may reflect menu design, marketing segmentation, or locked-program variety – not better hardware, better disclosure, or better outcomes. Preset count inflation is common: relabeling the same few underlying parameters into many named programs creates the appearance of versatility without adding transparency.
The more useful question is what operating parameters are visible and adjustable, not how many program names appear on the screen.
Why a Spec Disclosure Does Not Validate a Health Outcome
Disclosed specs can describe a device without validating a health outcome. Neither a number, a waveform label, nor a preset count proves treatment value. Outcome-proof logic is outside the scope of what any specification sheet can deliver.
Transparency is valuable for comparison. It is not valuable as medical evidence. These are separate categories, and collapsing them is the root cause of most overinterpretation in this market.
The Exact Point Where Description Becomes Overinterpretation
A PEMF claim starts as description when it names an operating value, control model, waveform, coil feature, or regulatory status. It becomes overinterpretation when the same sentence implies that the disclosed feature proves suitability, superiority, safety, or a health-related result. That transition is the main reading boundary buyers need to recognize.
In practical terms, the question is simple: does the sentence stop at what the device is, or does it continue into what the device supposedly does for the body? The first part may be comparison-useful. The second part often outruns what the disclosed spec can actually support.
How Condition-Led Reading Creates Unsupported Buying Logic
Reading specs through a condition lens – “which frequency helps with my joint pain?” or “which intensity is right for inflammation?” – turns descriptive data into implied therapeutic advice. This distorts comparison and crosses the boundary from buyer literacy into diagnostic or therapeutic substitution.
Stay with device descriptors and disclosure quality. The question is not “which spec matches my condition?” but “which device discloses its operating data clearly enough for me to compare it?”
Regulatory and Marketing Labels Buyers Should Classify Carefully
FDA labeling for PEMF devices follows administrative categories with specific meanings. Marketing labels often borrow the tone of regulatory authority without carrying its weight. Knowing the difference protects comparison integrity.
FDA Registered vs. FDA Cleared vs. Class Language
These phrases describe different regulatory situations and should be read carefully, not treated as interchangeable badges.
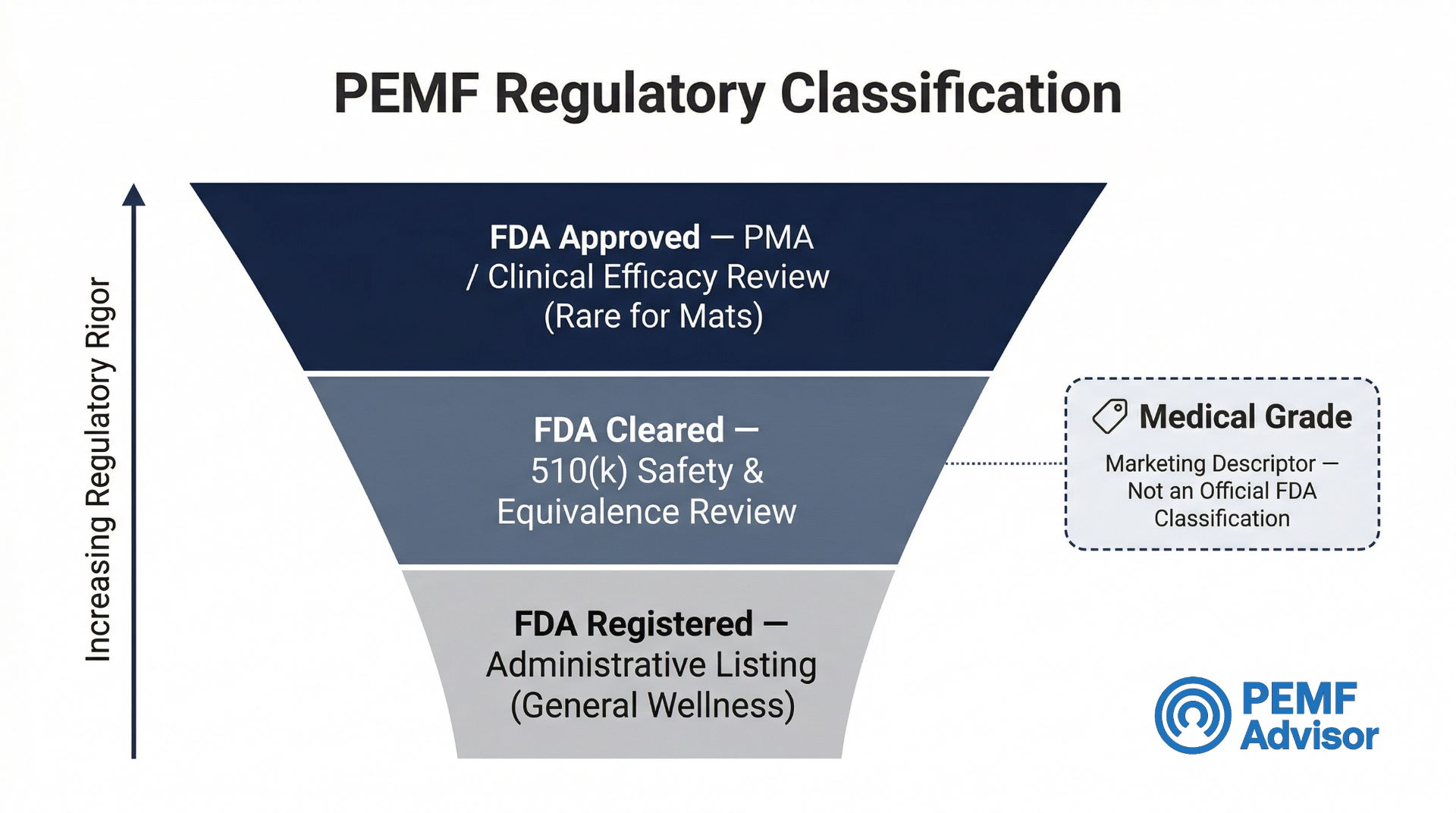
| Phrase | What It Means | What It Does NOT Mean |
| FDA Registered | The manufacturer has listed the establishment and/or device with the FDA | Does not mean the FDA reviewed, tested, or approved the device for medical use |
| FDA Cleared | The device has gone through a specific regulatory review pathway (e.g., 510(k)) | Does not mean the FDA endorses the device for specific diseases |
| Class I / Class II | Describes the regulatory risk category of the device | Should not be used as a shortcut to imply clinical proof or medical authority |
Why Registration Is an Administrative Listing, Not Disease Approval
FDA Registration for a PEMF mat is an administrative listing for a General Wellness device. It is not a medical approval for specific diseases. Registration should not be read as a proof-of-outcome label.
Conflating registration with clinical validation is one of the most common errors in PEMF marketing. When a product page prominently displays “FDA Registered” next to benefit claims, the two should be read separately: the registration describes an administrative status, and the benefit claim describes a marketing position. They do not reinforce each other.
A Fast Rule for Reading FDA Language on PEMF Product Pages
FDA wording should be read as a classification and administrative-status issue before it is read as a trust shortcut. “Registered” and “Cleared” describe different regulatory situations, and neither should be collapsed into a general feeling that the device’s operating claims are medically proven.
For buyers, the practical rule is simple: keep regulatory wording in its own category. Read it carefully, but do not let it silently validate surrounding benefit language that the page has not actually substantiated.
Why “Medical Grade” Is Not a Technical Spec for Home-Use PEMF Mats
“Medical Grade” is not a technical operating parameter in the home-use PEMF category. It does not function like Gauss, Hz, waveform disclosure, coil configuration, or control access. Unless tied to verifiable regulatory or technical context, it should be treated as marketing language.
Buyers often mistake this phrase for a specification because it sounds precise. It is not. No measurement, no comparison, and no regulatory pathway is invoked by the phrase on its own.
“NASA-Proven,” “Biocompatible,” and Similar Authority Shortcuts
Some labels borrow authority or scientific tone without improving comparability. “NASA-proven” may reference legitimate historical research, but it does not describe the device in your shopping cart. “Biocompatible” sounds technical but resolves into nothing you can compare against another product’s spec sheet.
These phrases should be classified cautiously unless they resolve into clear operating data or verifiable context. They may influence perception, but they do not improve your ability to compare one PEMF mat against another.
Trust and Corroboration Without Outcome-Proof Logic
Manufacturer transparency enables comparison of disclosed hardware data. Disclosure quality can improve trust in the comparison process. But transparency still does not prove biological or clinical outcomes. Missing details about measurement conditions, controls, or coils weaken comparison integrity – not because they hide proof, but because they reduce the information available for comparison.
Disclosure Quality as a Comparison Signal
Better disclosure lets you compare what is actually stated about the device: clarity on field strength, frequency behavior, waveform labeling, controls, and coils. Disclosure quality is useful because it reduces ambiguity – not because it validates results.
A product with thorough disclosure gives you more data points for comparison. A product with vague disclosure gives you less. The first is more useful for shopping; neither is more proven for health outcomes.
Transparency Helps Comparison More Than It Helps Certainty
Better transparency improves what the buyer can compare. It does not eliminate uncertainty about outcomes. A page that discloses more operating detail gives you a stronger basis for product-to-product reading, but it still does not turn a specification sheet into clinical proof.
This distinction matters because transparency often feels more authoritative than vague marketing. That feeling is understandable, but it should stay inside the comparison layer. Transparency reduces ambiguity. It does not remove the gap between device description and health effect.
What Better Transparency Looks Like
Stronger disclosure includes measurement context (where and how intensity was measured), clear frequency behavior (fixed value, range, or sweep pattern), standard waveform terms (not just proprietary labels), control visibility (what parameters can be adjusted), and coil information (count, material, arrangement).
Exact wording matters more than branding flair. A product page that says “800 Gauss at mat surface, copper coils, 1–50 Hz adjustable, sine wave” gives you more comparison material than one that says “powerful medical-grade PEMF with advanced healing programs.”
Disclosure Quality Checklist
- Is field strength stated with measurement location?
- Is frequency disclosed as a specific value or range?
- Are waveform labels standard or proprietary?
- Can you see the actual operating parameters behind presets?
- Is coil count, material, or layout disclosed?
- Is regulatory wording specific (Registered vs. Cleared)?
Missing Details That Weaken Comparison Integrity
Missing measurement location, incomplete intensity format, opaque presets, and absent coil disclosure all reduce comparison integrity. When detail is absent, you cannot verify whether two products are truly comparable or just similarly labeled.
The focus here is on what you cannot verify when data is missing. If a mat lists “3000 Gauss” but does not say where that was measured, you have a number without a frame. If another mat lists “500 Gauss at 4 inches from surface,” you have a smaller number with a more complete picture. The second is more useful for comparison despite the lower figure.
How to Treat Manufacturer Claims When the Data Sheet Is Vague
Vague data sheets should push you back toward classification, not inference. Broad promises without corresponding operating data should be read cautiously. When a product page is heavy on benefit language and light on measurable specs, the absence of detail is itself a signal.
The practical response is to notice what is missing and weight that absence in your comparison, not to fill the gap with assumptions about what the device can do.
What Belongs in Specs Versus What Belongs in Marketing
Some product-page elements belong to comparable operating data. Others belong to promotional framing and should be classified cautiously. Materials, heat, stones, and similar layers should be separated from PEMF operating data. The sorting principle is simple: compare directly, classify cautiously, and do not overread.
Specs Buyers Can Compare Directly
Field strength (Gauss/Tesla/µT), pulse rate (Hz), waveform labeling (square, sine, sawtooth), controls (preset vs. manual), coil disclosure (count, material, arrangement), and regulatory wording (Registered vs. Cleared) can all contribute to comparison when clearly stated. These are core descriptive fields, not proof fields.
Claims Buyers Should Read as Promotional Language
Labels such as “Medical Grade,” “NASA-proven,” cellular recharge language, and superiority wording should be treated as promotional unless supported by clear technical context. They may influence perception without improving comparability. When you encounter them, mentally move them to a separate category from the operating specs.
Hybrid Features That Should Be Separated from PEMF Operating Data
Stones, crystals, heat, fabrics, and layered materials are separate product features. They do not replace core PEMF operating-data disclosure. They may matter to ownership preferences – some buyers want far-infrared heat or amethyst layers for comfort or other reasons – but they do not function as PEMF hardware specifications.
When comparing PEMF mats, evaluate the PEMF specs on their own. Then evaluate bundled features as a separate layer. Mixing the two distorts comparison.
A Safe Buyer Checklist for Reading a PEMF Spec Page
When reading any PEMF product page, run through these five questions:
| # | Question to Ask | What You’re Looking For |
| 1 | What is measured? | Gauss, Hz, waveform label – specific numbers or terms, not benefit language |
| 2 | Under what conditions? | Measurement location, peak vs. average, distance from coil |
| 3 | What is adjustable? | Manual controls vs. locked presets; visible parameters vs. opaque programs |
| 4 | What is disclosed about hardware? | Coil count, material, arrangement – anything about the physical field-generation design |
| 5 | What is only promotional? | Labels like “Medical Grade,” “NASA-proven,” cellular language, superiority claims without data |
The goal is disciplined comparison, not therapy interpretation. Stick with device descriptors, notice what is missing, and keep promotional language in its own category.
The Minimum Reading Standard Before You Trust a PEMF Spec Claim
A PEMF spec claim becomes meaningfully useful when the buyer can identify at least three things: what the claim literally measures or describes, under what conditions that description is being presented, and whether the page stops at disclosure or keeps sliding into inferred outcome language. A fourth layer – how transparently the controller or hardware makes that claim verifiable – improves the comparison further but is not required to establish basic reading discipline.
This is not a verdict on product quality. It is a reading rule. The less clearly a claim separates device description from biological implication, the more cautious the buyer should become about treating it as serious comparison data.
FAQ
What counts as operating data on a PEMF product page?
Operating data covers measurable or directly disclosed device descriptors: field strength (Gauss/Tesla/µT), pulse rate (Hz), waveform labeling, control model (presets vs. manual), and hardware transparency (coil configuration). It excludes promotional interpretation, benefit claims, and marketing labels that cannot be measured or compared.
What does a Gauss number actually measure on a PEMF mat?
Gauss measures magnetic field strength. It is a technical intensity descriptor, not an outcome label. Context matters: the measurement location (surface vs. distance) and whether the figure represents a peak or average value both shape what the number actually tells you.
Why does distance matter when reading a PEMF intensity claim?
Field strength changes as distance from the source changes. A surface claim does not automatically describe the field farther away. Because intensity drops off quickly with distance, a headline Gauss number without measurement context is incomplete for comparison purposes.
What is the inverse square law in the context of PEMF mats?
The inverse square law is a physics principle describing how field strength drops quickly as distance from the source increases. For PEMF buyers, it means a Gauss number needs measurement context to be meaningful. It is a reading aid for interpreting spec claims, not treatment guidance.
What does Hz mean on a PEMF specification sheet?
Hz (Hertz) means pulses per second. It describes the pulse rate of the device and is part of operating data. It does not describe a condition-specific target, even if marketing materials frame certain frequencies as therapeutic.
What is the difference between a fixed frequency and a sweeping frequency range?
A fixed frequency stays at one stated pulse rate. A sweeping range changes across a stated interval or program cycle. This is a difference in operating behavior and disclosure type, not proof of different effects.
What does waveform geometry describe on a PEMF device?
Waveform geometry describes the pulse shape – labels such as square, sine, and sawtooth fall into this category. It is a descriptive specification about the electrical signal’s rise and fall pattern, not proof that one shape works better than another.
Does one waveform shape prove a PEMF mat works better?
No. Waveform shape alone does not prove better outcomes. Claims of superiority based on waveform should not be treated as established buying proof. The stable reading is descriptive only: the label tells you the pulse shape, not its biological effect.
What is the difference between presets and manual control on a PEMF mat?
Presets are pre-loaded programs that run fixed routines. Manual control means some operating parameters (frequency, intensity, or both) can be changed directly by the user. This difference describes control access and transparency, not treatment results.
Why does coil configuration matter as a disclosure-quality signal?
Coil configuration gives clues about how much hardware detail a manufacturer discloses. Better hardware transparency (coil count, material, arrangement) can improve comparability across products. It still does not prove health outcomes.
What is the difference between FDA Registered and FDA Cleared for PEMF mats?
These are different regulatory phrases. FDA Registered means the manufacturer listed the device with the FDA, typically as a General Wellness product. FDA Cleared means the device went through a specific review pathway such as 510(k). They should not be treated as interchangeable, and neither should be casually read as disease-proof language.
Does FDA registration prove a PEMF device is medically effective?
No. FDA registration is an administrative status, not a disease-specific efficacy verdict. It does not mean the FDA tested, approved, or endorsed the device for treating any medical condition. Buyers should not read registration as treatment proof.
Is “Medical Grade” a real technical specification for home-use PEMF mats?
No. “Medical Grade” is not a technical PEMF spec in this category. It does not function like Gauss, Hz, or waveform disclosure. Unless tied to verifiable regulatory or technical context, it should be treated as marketing language.
Do more presets prove a PEMF mat has greater healing potential?
No. More presets do not prove greater healing potential. Preset count reflects menu design or marketing segmentation, not outcome quality. Transparency around actual settings – visible parameters rather than named programs – is more useful for comparison.
Do crystals, heat, or layered materials count as PEMF operating data?
No. These are separate product features, not core PEMF operating parameters. They may be part of the product description but should be kept separate from field strength, pulse rate, and waveform disclosure when comparing PEMF specifications.
How should buyers treat manufacturer transparency when comparing PEMF mats?
Transparency can improve comparison quality. Buyers should look for clear operating data, measurement context, and hardware disclosure. However, transparency itself does not validate health outcomes. It makes comparison more reliable – that is its value.
Can a disclosed spec validate a health outcome?
No. A disclosed spec does not validate a health outcome on its own. Specs describe the device; they do not prove therapeutic results. Outcome-proof logic is outside what a specification sheet can deliver, regardless of how detailed the disclosure is.
What is the safest way to compare PEMF products without overinterpreting them?
Compare operating data (field strength, frequency, waveform, controls, coil disclosure) and regulatory wording. Classify promotional language separately. The goal is disciplined comparison without importing outcome-proof logic into device descriptions.

The PEMF Advisor Editorial Team reviews consumer PEMF mats and related wellness devices. Our work focuses on verified specifications, documentation, usability, materials, warranty/returns, and ownership considerations. We do not provide medical advice or evaluate health outcomes. See our Review Methodology and Editorial Standards.