PEMF specs on home-use devices are comparison tools, not proof of outcomes. To read a product page more clearly, focus on the fields that actually help you compare one device with another: frequency range, intensity, waveform, controller style, coil disclosure, and measurement context. This guide explains what each spec means, what it does not prove, and how to read controller language without relying on marketing shortcuts.
PEMF Advisor publishes buyer-oriented reviews, comparisons, and educational content for home-use PEMF mats and devices. This page is a technical literacy guide within that system: it explains how to interpret PEMF specifications and controller language for comparison purposes, but it does not provide medical advice, diagnosis, treatment guidance, or protocol recommendations.
If you need the broader category context before comparing individual specifications, start with our PEMF Buyer Education guide. That page explains the overall product category, format differences, bundled features, and transparency rules that sit around the narrower spec and controller questions covered here.
What Buyers Should Compare First in a PEMF Spec Sheet
The most useful order is simple: start with frequency range, magnetic intensity reporting method, waveform type, controller style, coil disclosure, and measurement transparency. Those fields create the technical comparison surface. They tell you what the device is willing to disclose about how it operates, not what it promises to accomplish.
This order matters because PEMF product pages often mix technical fields with persuasive language. A page can sound precise while still hiding the information that makes comparison possible. When the spec sheet gives you standard geometry names, measurement context, preset details, and hardware disclosure, you can compare products more objectively. When it gives you broad promises, proprietary labels, or undefined numbers, you are comparing marketing style more than device transparency.
PEMF Spec Comparison Table
Before diving into definitions, this table frames the core technical fields you will encounter on PEMF product pages. Each field serves as a transparency metric for comparison, not a medical proxy. How a manufacturer reports each field matters as much as the number itself.
| Spec Field | What It Describes | Comparison Use | Watch For |
| Frequency Range (Hz) | Cycles per second the device operates within | Shows operating range; wider is not automatically better | Claims linking specific Hz to conditions |
| Magnetic Intensity (Gauss/mT) | Magnetic flux density at a given point | Only comparable when measurement method and distance are disclosed | Peak vs. average not stated; measurement distance missing |
| Waveform Type | Pulse shape: square, sawtooth, sine, or proprietary | Describes signal geometry; does not validate outcomes | Proprietary names with no technical disclosure |
| Pulse Duration/Width | Timing detail within each pulse cycle | Adds precision to signal description when disclosed | Often omitted entirely from spec sheets |
| Controller Interface | How the user selects or changes settings | Differentiates preset-only vs. manual-control devices | Preset labels that sound medical without disclosure |
| Coil Count and Layout | Number and arrangement of electromagnetic coils | Useful only when layout, spacing, and zones are disclosed | Raw count without placement or coverage context |
Gauss vs. Tesla: Quick Conversion
1 Tesla = 10,000 Gauss. Home-use PEMF devices typically operate in the milliTesla (mT) or Gauss range. The unit language alone does not change the underlying measurement – always check how and where the reading was taken.
Use this table as a checklist: if a product spec sheet leaves any of these fields blank or undefined, the comparison value of that sheet drops. The presence of a number matters less than whether the manufacturer explains how that number was obtained.
Core Spec Fields Every Buyer Should Read
Four fields form the baseline for any product comparison: frequency range, magnetic intensity, waveform type, and controller type. If a spec sheet covers these clearly, you have enough to start comparing. Two additional fields – pulse duration and coil details – become useful when manufacturers disclose them with enough context.
These fields help you compare product descriptions. They do not predict clinical outcomes, and a spec sheet that covers all six is not automatically superior – it is simply more transparent and easier to evaluate against other products.
| Field | What to Look For |
| Frequency Range | Stated in Hz with low and high bounds |
| Magnetic Intensity | Value in Gauss or mT with measurement method noted |
| Waveform Type | Standard geometry name (square, sawtooth, sine) not just a brand name |
| Controller Type | Clear statement of preset-only, manual, or hybrid |
| Pulse Duration | Stated in microseconds or milliseconds when available |
| Coil Details | Count plus layout or zone description |
Fields That Often Appear Without Enough Context
Some specs can sound precise while remaining effectively unclear. This does not mean the specs are worthless – it means the disclosure around them is incomplete.
Gauss values without measurement context. A product listing “3,000 Gauss” tells you very little unless you also know where that reading was taken (at the coil surface, one inch away, or through a mat layer) and whether the figure represents peak output or an averaged reading. Two devices listing 3,000 and 1,500 Gauss might be closer in real-world output than they appear if one measures at the surface and the other measures at body distance.
Proprietary waveform names. When a manufacturer replaces standard geometry terms like square, sawtooth, or sine with a branded name but provides no underlying technical description, the label loses comparison value. Standard geometry names tell you the pulse shape. A proprietary name without disclosure does not.
Coil count without layout. A mat advertising “20 copper coils” sounds like more coverage than one with 12, but without knowing the spacing, zone arrangement, or field overlap, you cannot judge actual coverage. Layout context turns a raw count into usable information.
What a Transparent Spec Sheet Usually Includes
A transparent PEMF spec sheet usually does four things well. It names core fields in standard terms, it explains how the measurements were obtained, it shows what the controller can actually change, and it separates core PEMF variables from bundled or adjacent features. Those are not “bonus” details. They are what make one device easier to compare against another without filling the gaps with guesses.
At a minimum, a strong spec sheet should give you a stated frequency range, a magnetic intensity value with reporting context, a standard waveform name or clear technical equivalent, and enough controller disclosure to understand whether presets are transparent or opaque. If those elements are missing, the product may still be viable, but the comparison surface is weaker.
Transparency Checks That Make Two Similar-Looking Devices Easier to Compare
When two products list similar headline specs, the differences usually sit in places buyers do not think to check: measurement method, control flexibility, and hardware disclosure detail. The following checklist is not a scoring system – it is a way to identify which product gives you more usable information.
| Transparency Check | What It Tells You |
| Is the Gauss measurement distance stated? | Whether intensity figures can be compared at all |
| Is peak vs. average intensity specified? | Whether you are comparing the same type of number |
| Are waveform types named with standard geometry? | Whether the signal description is verifiable |
| Does the controller disclose which variables it changes? | Whether presets are transparent or opaque |
| Is coil layout described, not just coil count? | Whether coverage claims have hardware backing |
| Are regulatory references (FDA, FCC) given administrative context? | Whether safety labels are being misrepresented as treatment endorsements |
A product that satisfies more of these checks is not necessarily “better” in any clinical sense – it is more transparent, which means your comparison is built on disclosed information instead of assumptions.
What PEMF Specs Are For in Buyer Comparisons
PEMF specifications describe how a device operates. They do not describe what happens in your body when you use it. That distinction matters because much of the marketing around home-use PEMF devices blurs this line – presenting operating parameters as if they were clinical evidence.
Specs are transparency metrics. They tell you the frequency range a device covers, how strong its magnetic field is (and ideally, how that strength was measured), what shape the pulse takes, and how the controller lets you interact with those variables. When disclosed clearly, these fields let you compare two products side by side. When disclosed poorly, they become decoration.
Define Frequency, Intensity, Waveform, and Controller Fields
Frequency (Hz) refers to the number of pulse cycles per second. A device listing 1–99 Hz operates within that range of cycles. Frequency is an operating parameter – it describes the speed of signal repetition, not a treatment instruction.
Magnetic Intensity (Gauss or mT) measures magnetic flux density – essentially, how strong the magnetic field is at a given point. This value depends heavily on where and how the measurement was taken, which is why a standalone number without methodology context is incomplete.
Waveform Type describes the shape of the electromagnetic pulse. The three standard categories are square (sharp on/off transitions), sawtooth (gradual ramp with a sharp drop), and sine (smooth, continuous wave). Each describes geometry, not efficacy.
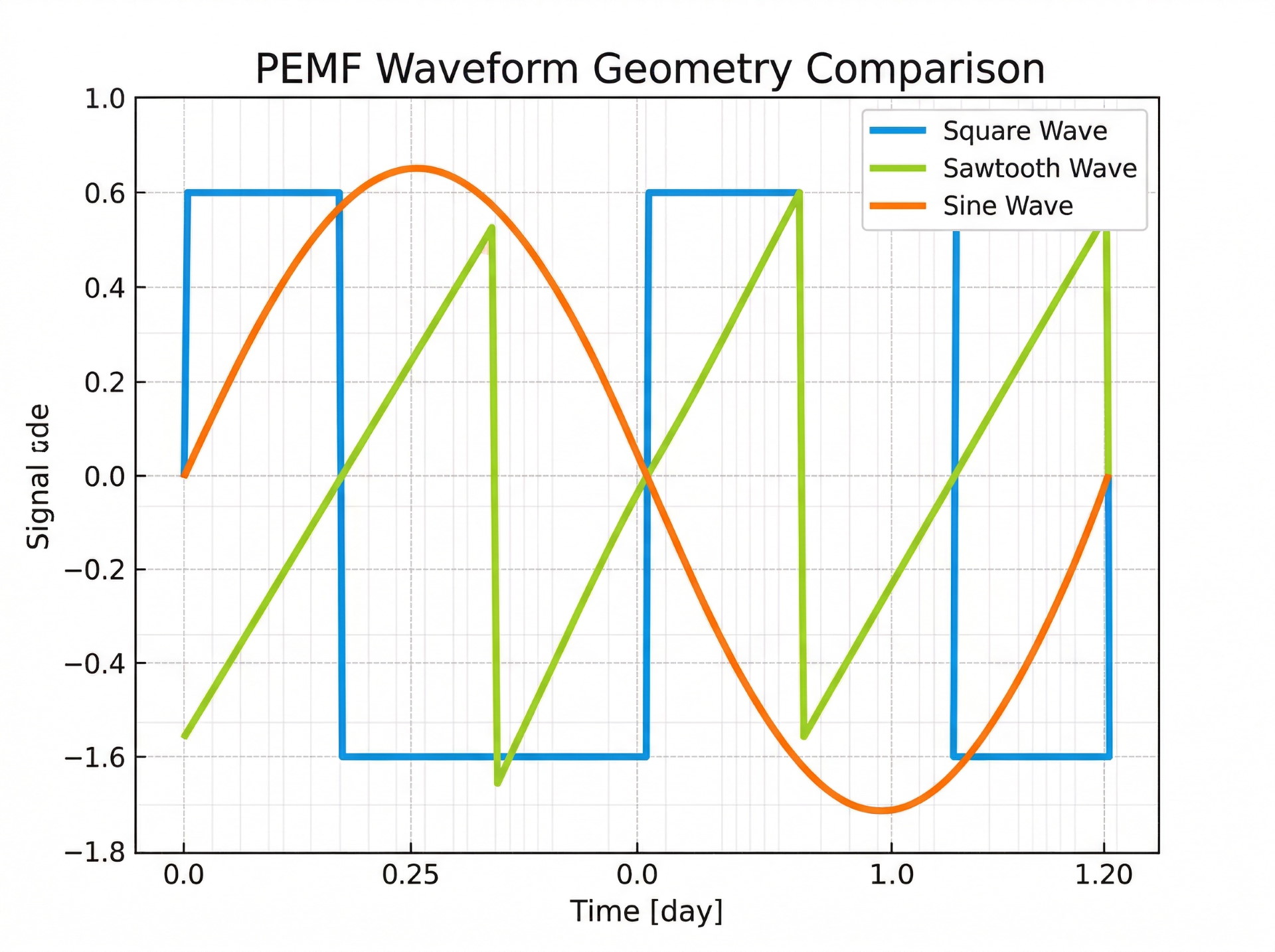
Controller Interface describes how the device lets you select or adjust settings. Some controllers offer only preset programs. Others expose manual controls for frequency, intensity, waveform, or timing. The controller determines what you can adjust, not what the adjustment will accomplish.
Separate Transparency Metrics from Performance Promises
| What Specs Measure | What Specs Do Not Prove |
| Operating frequency range | Treatment outcomes for any condition |
| Magnetic field strength at a stated point | Clinical superiority over other devices |
| Signal geometry (pulse shape) | That higher numbers mean better results |
| Controller flexibility and access | That presets target specific health goals |
| Hardware layout and coil arrangement | That a broader range equals broader benefit |
Some marketing materials claim that higher Gauss leads to faster tissue repair or that certain waveforms produce specific biological outcomes. These claims circulate widely in the home-use PEMF market, but they sit outside what technical specifications can validate on their own. Treat specs as description tools, not outcome guarantees.
Technical Reality vs. Marketing Interpretation
| Spec Layer | Marketing Shortcut | Technical Reality |
| Frequency | A specific Hz range is inherently tied to a specific condition | Frequency describes operating speed only; condition-matching claims exceed what the spec proves on its own |
| Intensity | Higher Gauss automatically means a stronger or better result | Gauss describes field strength at a stated point; meaning depends on distance and reporting method |
| Waveform | A specific waveform is inherently superior | Waveform describes signal shape; superiority claims remain unsettled |
| Controller | Presets prove the device targets specific wellness goals | Presets describe software organization unless the underlying settings are disclosed clearly |
A contrast like this keeps the distinction between measurable fields and interpretive claims visible. It does not say that all marketing language is false. It shows where comparison value ends and where unsupported interpretation begins.
Why Measurement Method Changes How Specs Look on Paper
Magnetic flux density decreases with distance from the coil. This follows predictable physics – the farther from the source, the weaker the reading. The practical problem for buyers is that two manufacturers can measure the same device and produce very different numbers depending on whether the reading was taken at the coil surface, at one inch, or through the mat’s padding layer.
On top of distance, there is a reporting-style variable. Peak intensity captures the maximum output during a single pulse cycle. Average intensity captures the mean output over time. A device reporting peak values will always display a higher number than one reporting averages – even if the actual output is identical.
Key Takeaway
When comparing Gauss or mT values across products, always ask two questions: Where was the measurement taken? Is the reported number peak or average? Without answers to both, the comparison has limited value.
Why Surface Measurements and Body-Distance Measurements Diverge
Magnetic intensity falls off as you move away from the coil. That means a reading taken at the coil surface will not match a reading taken through padding, through a cover layer, or at the distance where a body would normally sit relative to the hardware. Two published numbers can therefore describe the same category of device while representing very different measurement positions.
This is why distance disclosure matters as much as the headline number. A strong spec sheet tells you not only the reported Gauss or mT value, but also the measurement context that gives that value meaning. Without that context, the number is real but only partially interpretable.
How PEMF Controllers Work
The controller is the interface between you and the device’s hardware. It determines which variables you can see, adjust, or cycle through during a session. Understanding what a controller does – and what it does not do – helps you evaluate products based on usability and transparency rather than implied efficacy.
Presets vs. Manual Controls
Most home-use PEMF devices fall somewhere on a spectrum between fully preset-driven and fully manual. Preset controllers package pre-selected combinations of frequency, intensity, waveform, and timing into named programs. Manual controllers expose some or all of those variables directly to the user.
| Aspect | Preset-Heavy Controllers | Manual-Heavy Controllers |
| Usability | Simpler operation; choose a program, press start | Steeper learning curve; user selects each variable |
| Transparency | Low if preset contents are undisclosed | Higher when variable ranges are published |
| Flexibility | Limited to manufacturer’s program selections | Wider range of user-defined combinations |
| Risk of Misuse | Lower – fewer decisions for the user to make | Slightly higher if documentation is poor |
| Proof of Quality | Neither approach proves product quality on its own | Neither approach proves product quality on its own |
Manual control is not automatically superior to presets. A well-documented preset system that tells you exactly what each program changes can be more transparent than a manual interface with undocumented variable ranges.
What a Controller Can Change
Depending on the device design, a controller may expose any combination of the following variables: frequency selection within the hardware’s range, waveform type, pulse width or duration, session timing and auto-shutoff, and program sequencing (cycling through different settings within a single session).
A controller’s flexibility is always bounded by two constraints: the hardware it connects to and the software the manufacturer has built. A device with coils rated for 1–50 Hz cannot output 100 Hz regardless of what the controller allows you to input. Similarly, a locked software design may limit available waveform options even if the hardware could technically support more.
What a Controller Cannot Prove
A sophisticated controller with many adjustable variables does not validate the device’s therapeutic value. Preset labels like “deep relaxation” or “bone support” describe the manufacturer’s naming convention, not a verified medical targeting mechanism.
Some sources in the PEMF market claim that specific frequency presets or waveform geometries produce defined health outcomes. These claims are common in marketing materials but remain outside what controller design can independently verify. A controller is a usability tool. It is not clinical evidence.
Controller Transparency vs. Preset Opacity
Controller language becomes more useful when a manufacturer tells you exactly what the interface changes. A preset label such as “Relax” or “Recovery” has low comparison value on its own. It becomes more useful only when the product documentation explains which frequency range, intensity level, waveform type, or timing pattern sits behind that label.
The same rule applies to manual controls. A manual interface is only more transparent when the ranges are published clearly enough for a buyer to understand what the controls actually expose. A slider without a documented range is still opaque. In practical terms, the key question is not whether a device offers presets or manual control. The key question is whether the controller behavior is disclosed well enough to compare.
Example Comparison Scenario: Two Devices with Different Control Styles
Imagine comparing two home-use PEMF mats at similar price points:
| Feature | Device A | Device B |
| Controller Style | 8 locked presets, no manual adjustment | 3 presets plus manual frequency, intensity, and waveform selection |
| Preset Disclosure | Program names only (e.g., “Energy,” “Sleep”) | Each preset lists exact Hz, Gauss, waveform, and duration |
| Timer | Fixed at 30 minutes per program | Adjustable from 10 to 60 minutes |
| Coil Info | “Multi-coil design” with no layout detail | 12 coils in 3 zones with spacing diagram |
In this scenario, Device B provides more disclosed information. That does not make it clinically superior. It makes the comparison clearer. A buyer who values simplicity may still prefer Device A – but they should recognize that the spec sheet offers less transparency to work with.
First-Session Logic for Manual-Heavy Controllers
On a manual-heavy controller, the safest first comparison mindset is not “How should I optimize this?” It is “How much of this interface is actually disclosed?” Look for whether the device clearly states its available frequency range, intensity range, waveform options, timer logic, and any preset-to-manual relationship before assuming that more controls automatically create more value.
For buyers, the first-session question is usually a transparency question rather than a performance question. If the manual or interface does not make the settings legible, the extra control range may increase uncertainty rather than confidence. In that sense, usability begins with disclosed limits, not with the number of controls on the screen.
What PEMF Specs Can and Cannot Tell You
Each spec field describes one aspect of how a PEMF device operates. Together, they form a description layer – not a performance guarantee. This section breaks down what each field means in practice, where interpretation gets tricky, and where the limits of spec-based comparison sit.
Frequency Range as an Operating Parameter
Hertz (Hz) measures cycles per second. When a device lists a frequency range of 1–99 Hz, it means the device can operate at any speed within that band. The range describes available operating values or the boundaries of its programs.
Frequency range is not a condition-matching shortcut. Some marketing materials suggest that specific frequencies correspond to specific wellness goals, but these claims sit outside what a frequency specification can substantiate on its own. Use frequency range to understand how much variation a device offers, not to self-prescribe settings.
Gauss and Tesla as Field-Strength Reporting
Gauss and Tesla (or milliTesla, mT) are two unit systems for expressing magnetic flux density. They measure the same physical property – one Tesla equals 10,000 Gauss. Most home-use PEMF devices report in Gauss or mT because their output falls well below one full Tesla.
The reported value depends entirely on how and where it was measured. A device measured at the coil surface will show a higher number than one measured at three inches away. Neither number is wrong, but they are not directly comparable. Higher numbers alone do not establish product superiority – they may simply reflect a closer measurement point.
Waveform Geometry as Pulse Shape Description
The three standard waveform categories describe how the electromagnetic pulse rises, holds, and falls. Square waves have sharp on/off transitions. Sawtooth waves ramp gradually and drop sharply. Sine waves follow a smooth, continuous curve.
These categories describe signal shape – geometry, not guaranteed outcomes. When a manufacturer uses a proprietary waveform name instead of a standard geometry term and provides no underlying technical detail, the label should be treated cautiously. It may describe a real signal variation, but without disclosure, a buyer cannot verify that.
Some marketing claims describe square waves as the only effective waveform or use “NASA-proven” language as a blanket endorsement. These claims appear frequently in PEMF marketing but should not be treated as universal validation. NASA research involved specific protocols under specific conditions, and extending those findings to all square-wave home devices overstates what the research supports.
Pulse Duration and Sequencing as Control Detail
Pulse duration (or pulse width) describes how long each individual pulse lasts within a cycle, typically measured in microseconds or milliseconds. It adds timing precision to the signal description.
Sequencing refers to whether the device can cycle through different setting combinations during a single session – for example, starting at one frequency and waveform, then switching to another partway through. When disclosed, both fields improve the transparency and granularity of a product’s spec sheet. They do not, on their own, predict session outcomes.
Coil Count and Layout as Hardware Disclosure
The number of coils in a PEMF mat tells you very little without layout context. Twelve coils clustered in the center of a mat produce a different field pattern than twelve coils evenly distributed across three zones. Spacing, zone mapping, and field overlap all influence how the magnetic field distributes across the mat’s surface.
Coil transparency is a legitimate comparison field. A manufacturer that publishes a layout diagram or describes zone coverage gives you hardware information you can actually use. A manufacturer that lists only “20 coils” without placement detail does not.
Distance, Peak vs. Average, and Reporting Variability
This is where most spec-sheet confusion originates. Magnetic intensity decreases as distance from the coil increases – following physics, not opinion. A device reading 3,000 Gauss at the coil surface might measure 800 Gauss at two inches. Both readings are accurate. Neither is the “real” number in isolation.
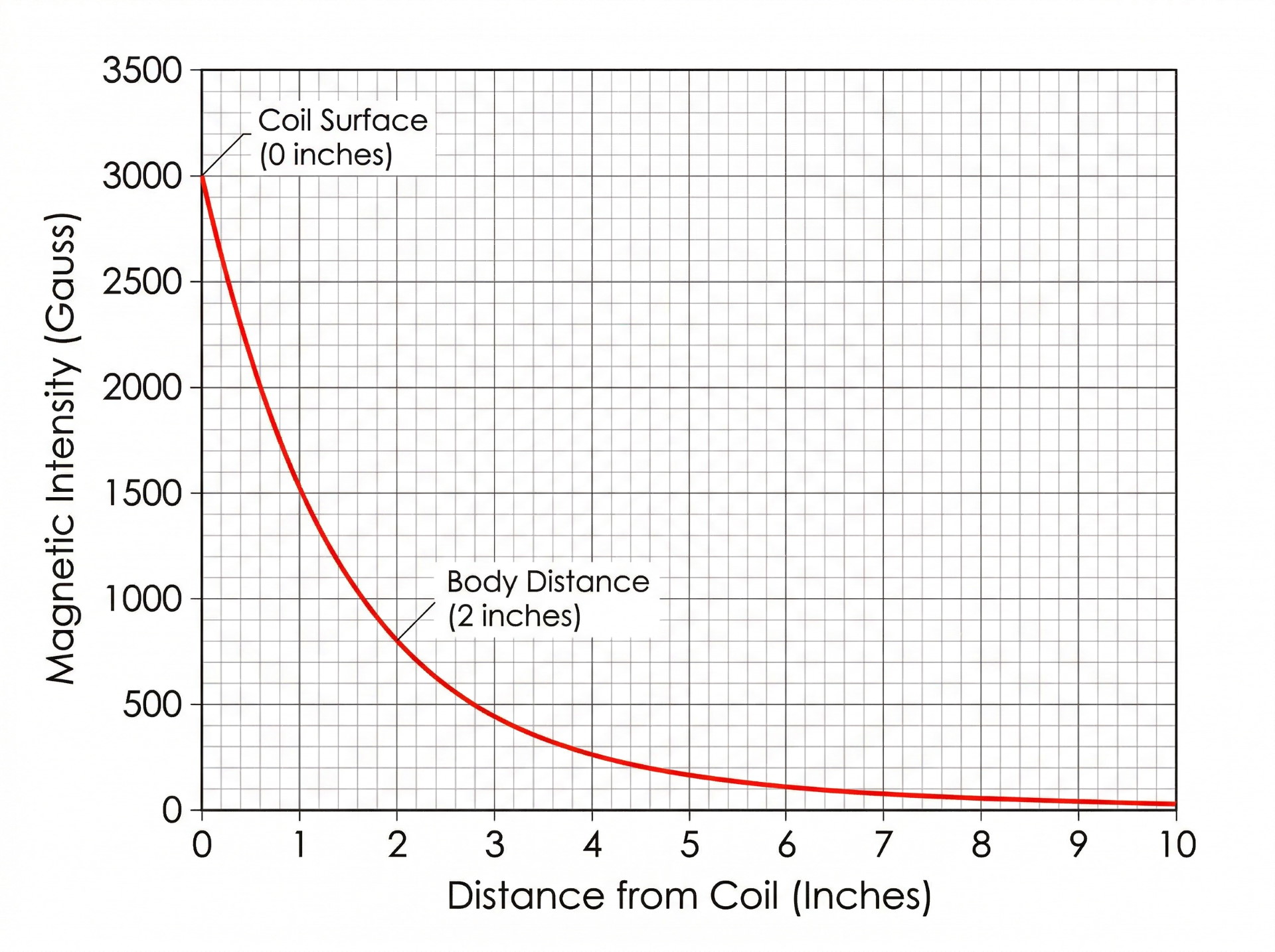
On top of distance, the reporting choice between peak and average values further changes how products appear on paper. Peak captures the maximum output during a pulse. Average captures the mean over the full cycle. A device reporting peak values will always display a higher number than one reporting averages, even with identical hardware.
The Buyer’s Rule
Any time you compare Gauss or mT across two products, treat the numbers as incomplete until you know: (1) the measurement distance, and (2) whether the figure is peak or average. Without both, you are comparing labels, not output.
How to Compare Disclosure Quality Across Home-Use PEMF Devices
Disclosure quality is the practical lens that separates a usable spec sheet from a decorative one. When two devices look similar on the surface, the depth and clarity of what each manufacturer discloses is what determines whether you can make an informed comparison.
Measurement Methodology and Distance Disclosure
When evaluating a Gauss or mT claim, look for answers to three questions: Where was the reading taken? What instrument was used? Is the number peak or average? If the product page or manual does not address at least the first and third, the intensity spec has limited comparison value.
Missing methodology does not necessarily mean the manufacturer is hiding something. It may mean the spec sheet was written for marketing rather than technical comparison. Either way, the absence of measurement context weakens the number’s usefulness to you as a buyer.
Coil Design Transparency and Hardware Details
Ask whether the manufacturer discloses coil placement, spacing between coils, zone coverage, and any shielding details. Coil shielding (EMI or EMF protection) may appear as a supporting detail – it is relevant to build quality, but it should not distract from the more fundamental question of how the magnetic field distributes across the mat.
A spec sheet that describes coil layout with a diagram or zone explanation provides genuinely more hardware transparency than one listing only a coil count. This is a comparison field, not a quality verdict – but it tells you which manufacturer is giving you more to work with.
Manual Programmability vs. Locked Presets
In the context of disclosure quality, the question is not whether manual is better than presets. The question is whether the manufacturer tells you what the controller does. A preset system that publishes the exact parameters of each program is more transparent than a manual system that gives you sliders but does not document their ranges.
Flexibility and transparency are separate qualities. A device can be highly flexible but poorly documented, or rigidly preset but fully disclosed. Neither controller style alone proves product quality.
Power Source, Timer Logic, and Ownership Context
These are practical ownership fields that most buyers overlook until after purchase. Internal versus external power design affects portability and session logistics. Timer and auto-shutoff logic determines how sessions are managed and whether you need to actively monitor duration.
None of these fields prove outcomes. They affect convenience, safety behavior, and daily use patterns – which matters for long-term ownership satisfaction even though it is not a clinical consideration.
Regulatory and Safety Labeling as Descriptive Context
PEMF devices may be referenced in FDA or FCC contexts, but these references require careful interpretation. FDA “registration” for a device is an administrative process. It does not mean the FDA has approved the device for specific treatment claims. Registration and approval are different administrative categories.
When a product page states “FDA registered,” read it as an administrative filing, not a clinical endorsement. The same applies to FCC references, which relate to electromagnetic emissions compliance, not health outcomes. Treating regulatory labels as treatment validation is a common misread that spec-sheet transparency should help prevent.
Marketing Red Flags and Low-Trust Claims
Certain patterns in PEMF marketing consistently signal low disclosure quality. Recognizing them does not mean the product is bad – it means the marketing is not giving you enough information to judge.
| Red Flag Pattern | Why It Reduces Trust |
| “NASA-proven” as a blanket claim | NASA research involved specific protocols; extending findings to all devices overgeneralizes |
| Proprietary waveform name, no technical detail | Cannot verify the signal or compare it to standard waveforms |
| “Higher Gauss = faster healing” | Conflates field strength with clinical outcome without evidence context |
| Waveform linked to specific organ targeting | Signal shape describes geometry, not tissue-specific action |
| “FDA approved” (when only registered) | Misrepresents administrative status as clinical endorsement |
The presence of these patterns does not automatically disqualify a product. It does mean the spec sheet is leaning on marketing language rather than disclosed technical detail – and that should lower your confidence in comparing it objectively against more transparent alternatives.
Minimum Disclosure Threshold Before You Trust a Spec Sheet
If a manufacturer leaves multiple core comparison fields undefined, your confidence in the spec sheet should drop. A useful mental threshold is simple: if the page does not clearly disclose waveform type, measurement context for intensity, controller behavior, or coil layout logic, pause before treating the product as easy to compare.
This is not a hard rejection rule and it is not a verdict on product quality. It is a comparison-discipline rule. The more blank or weakly defined core fields a spec sheet contains, the more of the comparison is being carried by interpretation instead of disclosure.
FAQ
What does Hz mean on a PEMF device?
Hz stands for Hertz and measures cycles per second. On a PEMF device, it describes how many pulse cycles the device produces each second. It is an operating parameter, not a treatment instruction.
What is the difference between Gauss and Tesla?
Both are units for measuring magnetic flux density. One Tesla equals 10,000 Gauss. Home-use PEMF devices typically report in Gauss or milliTesla (mT). The unit choice does not change the underlying measurement – it is simply a different scale, like Fahrenheit and Celsius for temperature.
Does a higher Gauss number automatically mean a better PEMF device?
No. A higher Gauss number may reflect a closer measurement point, peak-only reporting, or a genuinely stronger output – you cannot tell which without knowing the measurement distance and reporting method. Disclosure quality around the number matters more than the number itself.
Why can two PEMF devices list very different Gauss numbers?
Because manufacturers may measure at different distances from the coil and use different reporting methods (peak vs. average). Two devices with identical hardware could produce different published numbers simply because one measures at the surface and the other at body distance.
What does waveform mean on a PEMF controller?
Waveform describes the shape of the electromagnetic pulse the device produces. The three standard categories are square (sharp transitions), sawtooth (gradual ramp, sharp drop), and sine (smooth curve). These describe signal geometry, not treatment outcomes.
Are square, sawtooth, and sine waveforms proof of better outcomes?
No. Waveform categories describe pulse shape. Claims tying specific waveform geometry to guaranteed health results are common in marketing but are not substantiated by the technical specification itself.
What is pulse duration or pulse width on a PEMF device?
Pulse duration (or pulse width) describes how long each individual pulse lasts, usually measured in microseconds or milliseconds. It adds timing detail to the signal description and improves spec transparency when disclosed, but it is not an outcome guarantee.
What does a PEMF controller preset actually change?
A preset may adjust one or more variables such as frequency, intensity, waveform, pulse timing, or session duration, depending on the manufacturer’s design. Preset labels describe the manufacturer’s naming convention – they do not prove medical targeting.
Is manual control always better than presets?
Not necessarily. The better fit depends on how much variable control you actually want, how well the manufacturer documents each option, and your comfort level with adjusting technical settings. A well-disclosed preset system can be more transparent than a poorly documented manual interface.
Why does coil count matter only when the manufacturer explains the layout?
Because a raw coil count without placement, spacing, or zone context does not tell you how the magnetic field distributes across the mat. Twelve coils clustered in one area produce a different coverage pattern than twelve coils spread across three zones.
Why is distance from the coil important when reading magnetic intensity claims?
Magnetic field strength decreases as distance from the coil increases. A reading taken at the coil surface will be significantly higher than one taken at body distance. Without knowing the measurement distance, you cannot fairly compare intensity claims between products.
What is the difference between peak intensity and average intensity?
Peak intensity captures the maximum output during a single pulse cycle. Average intensity captures the mean output over the full cycle. These are different reporting methods – substituting one for the other in a direct comparison produces misleading results.
Does FDA registration mean a PEMF mat is approved for treatment claims?
No. FDA registration is an administrative process – it means the manufacturer has listed the product with the FDA. It does not mean the FDA has evaluated or approved the device for specific treatment claims. Registration and approval are different categories.
What should buyers do with proprietary waveform names that have no technical disclosure?
Treat them cautiously. A proprietary name without underlying technical detail cannot be compared to standard waveform categories. If the manufacturer does not explain the signal geometry behind the name, the label has limited comparison value. Standard terms like square, sawtooth, and sine are more useful for product-to-product evaluation.
What are the most useful transparency checks when comparing home-use PEMF devices?
Look for: (1) measurement method and distance for intensity claims, (2) standard waveform geometry names instead of proprietary labels, (3) controller documentation showing what each preset or setting changes, (4) coil layout detail beyond a raw count, (5) pulse duration disclosure, and (6) regulatory references placed in administrative context rather than presented as treatment endorsements. These checks help you compare on disclosed information rather than marketing language.

The PEMF Advisor Editorial Team reviews consumer PEMF mats and related wellness devices. Our work focuses on verified specifications, documentation, usability, materials, warranty/returns, and ownership considerations. We do not provide medical advice or evaluate health outcomes. See our Review Methodology and Editorial Standards.