Quick answer: We verify PEMF product information by checking what a brand actually discloses, where that information appears, whether it stays consistent across product materials, and whether the claim can be interpreted as a product fact rather than a treatment promise. We can confirm many product details as buyer-facing information. We do not treat that process as clinical validation or proof of health outcomes.
PEMF product pages often mix real specifications with broad language that sounds more certain than it is. A page may clearly disclose a controller, a frequency range, a warranty term, or a return window, then place those details beside claims that are much harder to verify in the same way. That is where verification matters. This page explains how PEMF Advisor checks product claims and product information before using them in reviews, comparisons, and buyer guides, and where that process stops.
What this page is for
This page explains what we mean when we say we verify PEMF product information.
That does not mean we run clinical trials. It does not mean we certify health outcomes. It does not mean we independently prove that a device delivers a therapeutic result for a specific user or condition.
It means something narrower and more useful for buyers.
We check whether a product claim is being presented as:
- a clearly disclosed product fact
- an interpretable technical statement
- a policy or ownership term
- a regulatory or administrative label
- a broad claim that exceeds what the available product evidence can responsibly support
That distinction matters because buyers often need help with a simpler question than the market suggests. They do not only need to know what a brand says. They need to know what kind of statement it is.
The shortest version of our verification model
We verify product information in layers.
First, we check whether the claim is about something that can actually be checked in buyer-facing product materials. Then we check whether the brand discloses it clearly enough to interpret. Then we check whether the same information stays consistent across the product page, specifications, manuals, policy pages, and other relevant sources.
If the information is consistent and clearly presented, we may treat it as verified product information in buyer terms.
If the information is vague, unsupported, unstable, or framed as more than the evidence really supports, we may classify it more cautiously.
That is the core model:
disclosed, interpretable, consistent, and bounded beats dramatic but unclear.
What we are actually trying to verify
We verify product-facing facts first
The strongest verification targets are the things a buyer can reasonably expect a publisher to check as product information.
That includes areas such as:
- product format and category
- controller type
- named settings and presets
- disclosed frequency ranges
- disclosed intensity language and measurement context
- waveform naming, if provided
- product dimensions and weight
- warranty terms
- return and refund conditions
- trial periods
- support or documentation availability
- regulatory wording used on the product page
These are not all equal in strength, but they are all part of the buyer-facing evidence layer.
We verify disclosure, not mythology
Some PEMF product pages include long chains of implied authority. A brand may combine technical terms, broad wellness language, regulatory references, historical references, and emotional framing in a way that makes the whole page feel proven.
Verification cuts through that effect by asking simpler questions:
- What exactly is being claimed?
- Where is the claim stated?
- Is it specific enough to interpret?
- Is it repeated consistently?
- Can it be checked as product information?
- Does it go beyond what the product evidence actually supports?
That is why our process focuses on the structure of the claim, not just its tone.
What we can usually verify with reasonable confidence
Product structure and format
We can usually verify what kind of product is being presented.
For example, we can often confirm whether a device is being sold as:
- a full-body mat
- a portable mat or pad
- a localized applicator
- a hybrid stacked product with PEMF plus other features
- a simpler core PEMF device
- a controller-led or preset-led system
This kind of verification helps buyers classify what they are looking at before they start comparing claims inside the product page.
Product specifications as disclosed
We can often verify that a brand discloses certain specifications.
That is an important distinction. Verification here does not mean we automatically prove the best possible interpretation of the number. It means we can confirm that the product page or supporting materials disclose a spec in a specific way.
For example, we may be able to confirm that a brand discloses:
- a frequency range
- an intensity number
- a waveform name
- a controller setting structure
- a timing or session-control feature
- a coil count
- a dimensions or weight spec
That is useful because buyers first need to know what is being disclosed at all, and only then whether the disclosure is strong, weak, or incomplete.
Policy and ownership terms
Warranty windows, return conditions, refund language, and trial offers are often among the most useful claims to verify because they affect buying risk directly.
These claims are also easier to check in a bounded way than broader wellness language. A return policy is either disclosed clearly or it is not. A warranty term is either stated with usable conditions or it is not. A support page either clarifies ownership burden or leaves too much hidden.
That makes policy verification a meaningful part of product evaluation.
Regulatory and administrative wording
We can also verify how regulatory or administrative language is being used.
For example, we may check whether a page uses words like:
- registered
- cleared
- compliant
- non-ionizing
- medical-grade
- wellness device
- professional
But verification here means we are checking the wording, the context, and the buyer meaning of the wording. It does not mean we turn those labels into medical proof.
What we treat more cautiously
Some claims are much harder to verify responsibly in a buyer-facing publishing context.
Broad outcome claims
If a claim moves from product description into strong outcome language, verification becomes much narrower.
A buyer-facing publisher can often verify that the claim exists. That does not mean the publisher can verify the outcome itself.
This is where the distinction matters most:
- we may verify that a product page says something
- we may classify the claim as broad, unstable, or overread
- we may refuse to treat it as settled product evidence
That is not hesitation for its own sake. It is part of keeping the page trustworthy.
Claims that collapse multiple ideas into one shortcut
Some PEMF claims sound strong because they merge several different layers into a single conclusion.
For example:
- a measurable field becomes a treatment implication
- a technical term becomes a trust shortcut
- a historical reference becomes a modern proof claim
- a regulatory phrase becomes a quality verdict
- a stacked feature becomes evidence that the PEMF system is stronger
These are the kinds of claims we slow down and classify carefully instead of repeating at face value.
Claims that exceed buyer-facing evidence
A manufacturer may imply something that goes beyond what the product page, documentation, or disclosed materials actually support.
In those cases, our role is not to dramatize the claim. Our role is to identify the gap between:
- what is being suggested
and - what is actually disclosed clearly enough to verify as product information
The main claim types we work with
The table below shows the practical difference between what we can usually check, what we can often classify, and what we do not treat as confirmed proof.
| Claim or Information Type | How We Check It | What We Can Confirm | What We Cannot Conclude |
|---|---|---|---|
| Product format | Product page, specs, images, manuals, category language | What kind of product is being sold | That format is automatically better for outcomes |
| Frequency disclosure | Product specs, controller screenshots, manuals | That a range or setting is disclosed | That the range proves a specific result |
| Intensity language | Spec sheets, measurement wording, product pages | That a number is disclosed and how it is framed | That the number alone proves superiority |
| Controller behavior | Product pages, manuals, interface descriptions | Whether settings are manual, fixed, preset-led, or adjustable | That one control style is universally better |
| Warranty and return terms | Policy pages, checkout disclosures, support docs | What the brand states as policy | That the ownership experience will be friction-free |
| Regulatory wording | Product pages, compliance statements, support materials | How the brand uses those terms | That the wording proves clinical effectiveness |
| Feature stacking | Product page layout, material descriptions, spec blocks | Whether PEMF is combined with heat, infrared, gemstones, or other features | That stacked features improve PEMF performance |
| Broad wellness claims | Product pages, marketing copy, FAQs | That the claim exists and how it is framed | That the outcome itself is proven by the product page |
How we actually check product information
We start with primary product materials
The first source is usually the product page itself. That is where the brand is choosing how to present the product to a buyer.
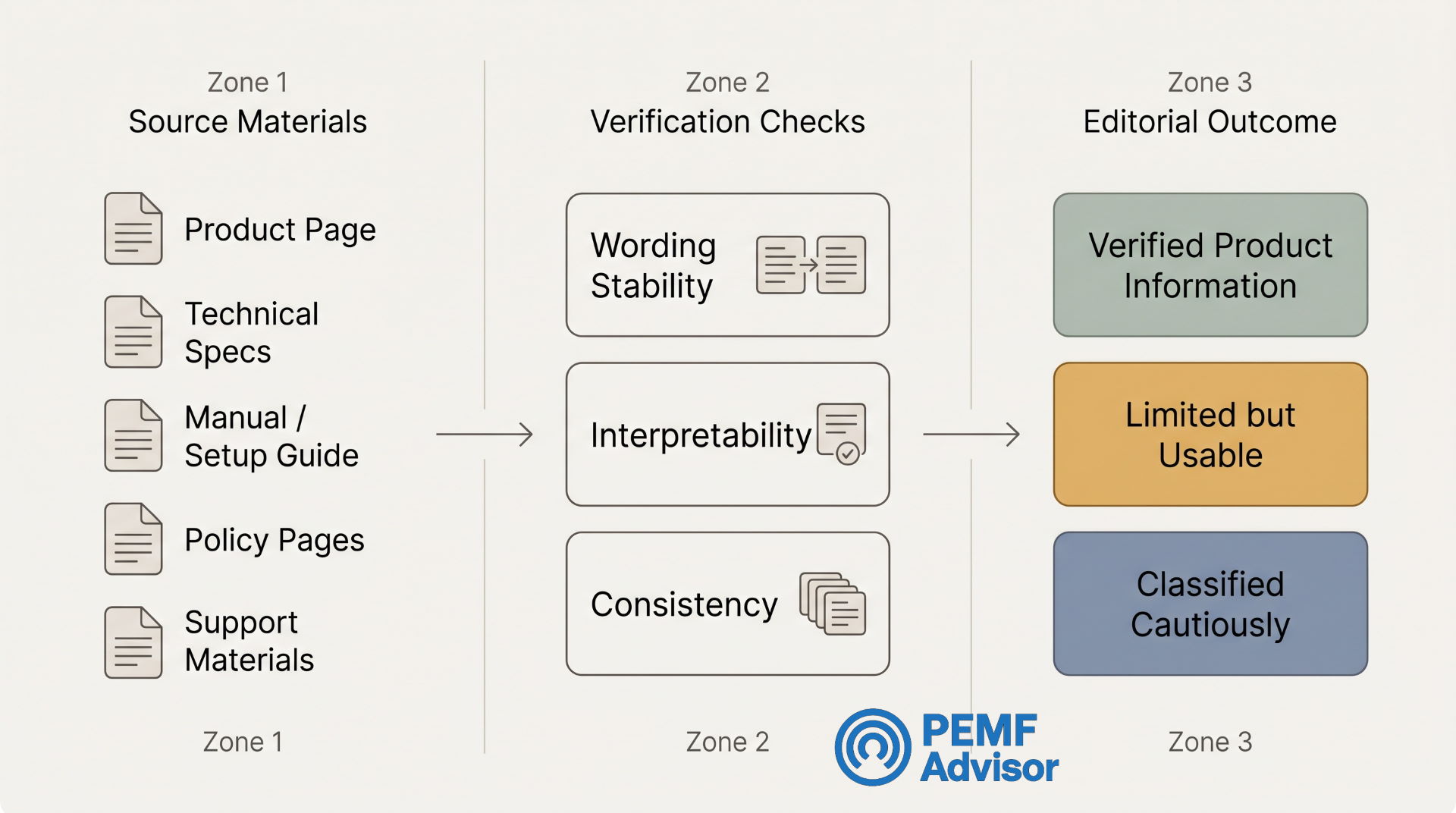
But we do not stop at the headline section. A claim may sound different depending on where it appears:
- in the hero section
- in the technical specs
- in the FAQ
- in the controller description
- in the warranty or support pages
- in downloadable manuals or setup guides
A claim that appears clearly and consistently across those layers is stronger than a claim that appears once in a dramatic headline and then disappears when a buyer looks for specifics.
We check whether the wording stays stable
Consistency matters.
If a product page suggests one thing, the specs imply another, and the support materials leave the issue vague, that weakens the claim from a buyer-verification standpoint. A product does not become easier to trust because it uses more pages. It becomes easier to trust when those pages tell the same story.
That is why we check whether a disclosed fact remains stable across the brand’s own materials.
We check whether the claim is interpretable
Some claims are technically present but still too unclear to help a buyer.
For example, a brand may disclose a field-strength number without enough context to compare it properly. Or it may mention a proprietary mode without explaining what the user can actually change. Or it may use a waveform label without enough surrounding explanation to make it meaningful.
In those cases, the issue is not only whether the claim exists. The issue is whether a buyer can interpret it honestly.
We check whether the claim belongs to the product layer at all
Not every statement on a product page belongs to product verification.
Some belong to:
- branding
- mood-setting
- emotional positioning
- adjacent lifestyle framing
- broad wellness storytelling
Those may still influence perception, but they are not the same kind of thing as a disclosed frequency range, a controller interface, or a return policy.
Part of verification is separating:
- product information
from - impression language
What we do when the claim is weak, vague, or inflated
We may classify rather than endorse
A weak claim does not have to be repeated as if it were a stable fact.
Sometimes the right move is to classify it instead:
- descriptive but limited
- disclosed but weakly interpretable
- marketing-forward
- context-dependent
- unsupported as stronger inference
- outside the product-verification layer
That gives the buyer a more honest reading than either blindly repeating the claim or refusing to mention it at all.
We may narrow the claim back to what is actually supportable
Often the best verification move is not to reject the whole statement. It is to pull it back to the part that can actually be supported.
For example, a dramatic product claim may shrink to:
- a disclosed spec
- a product-design difference
- a controller feature
- a policy term
- a descriptive material choice
That kind of narrowing is useful because it keeps the review grounded in what the buyer can actually compare.
What this process does not do
This process has clear boundaries.
It does not clinically validate outcomes
We do not use this page to certify that a PEMF product delivers a therapeutic result for a condition, a symptom, or a specific user type.
That is outside the role of this site.
It does not replace independent lab testing we do not have
If a claim would require controlled measurement, specialist equipment, or formal lab conditions to confirm properly, we do not pretend that normal buyer-facing publishing has already completed that work.
We may discuss what the brand discloses. We may assess how interpretable the disclosure is. We do not dress that up as independent proof if it is not.
It does not turn product verification into medical authority
This site evaluates home-use PEMF products as products. It does not provide diagnostic guidance, treatment instructions, or clinical interpretation. That is not a disclaimer added at the end. It is part of how the whole verification process is designed.
Why this matters for readers
Verification is not only about catching bad claims. It is about making comparison more readable.
A buyer in this category usually has to sort through three different layers at once:
- the product itself
- the way the product is described
- the larger story the brand wants the buyer to believe
Those layers are not always aligned.
A good verification process helps readers answer simpler and more practical questions:
- Is this a real disclosed product fact?
- Is the wording clear enough to interpret?
- Is this policy term actually visible and usable?
- Is this a technical statement or a marketing shortcut?
- Is this a claim we can treat as bounded product information, or only as brand language?
That kind of clarity is often more useful than a louder headline.
How this page supports the rest of the site
This page supports reviews, comparisons, rankings, and buyer guides by making the verification layer visible.
It explains why some claims appear in our coverage as:
- confirmed product disclosures
- product details with context
- buyer-useful but limited facts
- weakly interpretable marketing language
- claims that are classified rather than trusted
That is important because product evaluation becomes stronger when readers can see not only what we say, but how we decide what deserves confidence.
FAQ
Does PEMF Advisor independently prove that PEMF products deliver health outcomes?
No. This page explains how we verify product claims and product information in buyer-facing terms. It does not clinically validate treatment outcomes or therapeutic effectiveness.
What kinds of claims can you usually verify most clearly?
Product format, controller features, disclosed specifications, warranty terms, return policies, support details, and the way regulatory or administrative wording is used on the product page.
What does it mean when a claim is classified rather than trusted?
It means the claim may exist on the product page, but the available buyer-facing evidence does not justify treating it as a stable, strongly supported fact in the way the brand implies.
Do you verify every number on a product page as laboratory truth?
No. We verify how the number is disclosed, how it is framed, and whether it is interpretable in buyer terms. That is different from independently reproducing every measurement under lab conditions.
Why do you spend so much time on wording and context?
Because in this category, wording often does more work than buyers realize. A claim can sound strong while still being too vague, too context-dependent, or too broad to support the conclusion being suggested.
Do regulatory labels count as proof that a product is clinically effective?
No. We may verify how a label or phrase is used, but we do not treat regulatory or administrative wording as automatic proof of treatment effectiveness.
Does this page apply to reviews and comparisons too?
Yes. This verification framework supports the way product information is handled across reviews, comparisons, and buyer guides on the site.
Why not just repeat what manufacturers say and let buyers decide?
Because buyers are usually already seeing the manufacturer’s version. The publisher’s job is to separate clear product information from inflated interpretation so the comparison becomes easier to trust.

The PEMF Advisor Editorial Team reviews consumer PEMF mats and related wellness devices. Our work focuses on verified specifications, documentation, usability, materials, warranty/returns, and ownership considerations. We do not provide medical advice or evaluate health outcomes. See our Review Methodology and Editorial Standards.